Feature Article
Flu Shots Are Especially Important This Year
Getting a flu shot is recommended every year for individuals 6 months or older. During the COVID-19 pandemic it is even more important. The two viruses are expected to circulate simultaneously and for now protecting ourselves against the flu is important to prevent hospitalization from the flu as well as to reduce the risk of having both infections at the same time.
Are They Safe?
Flu vaccines are considered generally safe and well-tolerated, but they are associated with numerous flu-like and other side effects.
Are They Effective?
The overall effectiveness of annual flu vaccines depends mainly on how well the flu strains in the vaccines match the strains in current circulation. As the flu season is just beginning, this years’ vaccine effectiveness estimates will be provided when they become available. Recent studies of past vaccines effectiveness have shown that the vaccine reduces the risk of flu illness by about 40-60% among the overall population during seasons when most circulating flu viruses are like the vaccine viruses.
In a 2017 study, researchers showed that the flu vaccination reduced deaths by 52-79%, intensive care unit (ICU) admissions by 37%, ICU length of stay, and overall duration of hospitalization among hospitalized adults with the flu. In a 2018 study, researchers showed that a vaccinated adult who was hospitalized with the flu was 59% less likely to be admitted to the ICU and spent four fewer day in the hospital than those who had not been vaccinated.
“In a 2017 study, researchers showed that the flu vaccination reduced deaths by 52-79%.”
Clinical Infectious Diseases (CID)
When and What Vaccine Should I Get?
Manufacturers have already begun distributing flu vaccines and the Centers for Disease Control and Prevention (CDC) and the Advisory Committee on Immunization Practices (ACIP) recommend getting a flu vaccination as soon as possible. Private Health Management recommends all our clients receive the quadrivalent vaccine if available. The best time to get the vaccine this year for maximum protection was during October. If you missed that window, you should still get one as soon as possible.
The vaccine options for this 2020-2021 Flu Season include:
- One high dose quadrivalent flu shot for people 65 years of age and older
- Two flu shots using an adjuvant (an ingredient that helps create a stronger immune response), approved for people 65 years of age and older
- Several standard dosed quadrivalent flu shots, which are approved for children as young as 6 months of age
- There is also a quadrivalent cell-based influenza shot contains virus grown in a cell culture, which makes this vaccine totally egg-free. This is approved for people aged 4 years and older.
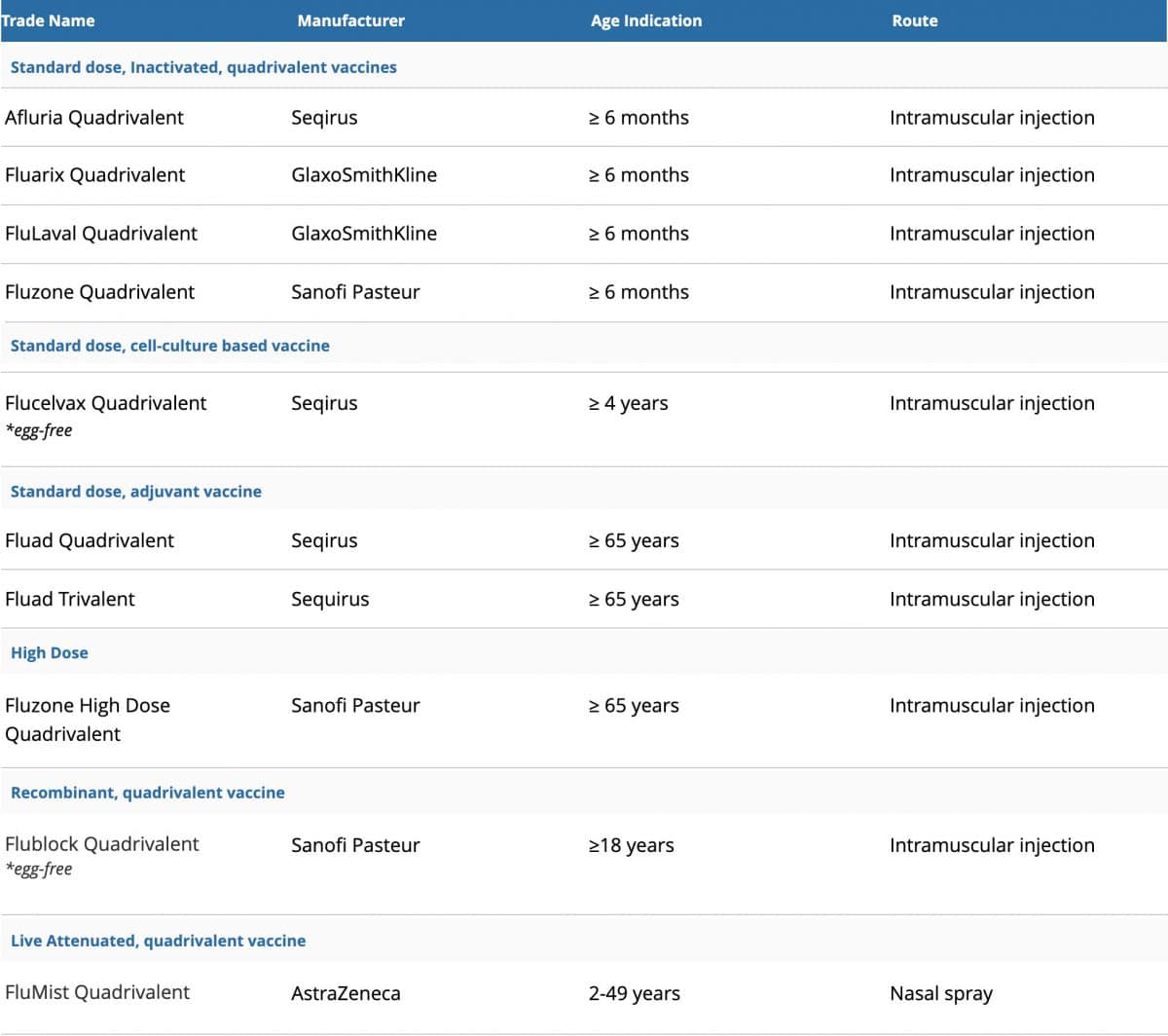
*Trivalent vaccine protects against three flu viruses: the two most common A strains (H1N1 and H3N2) and one B strain (Victoria)
**Quadrivalent vaccines protect against four flu strains: the same three viruses in the trivalent vaccine and an additional B strain (Yamagata)
CDC website: https://www.cdc.gov/flu/season/faq-flu-season-2020-2021.htm
